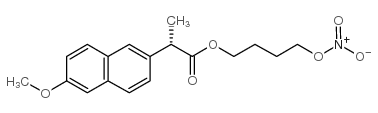
NAPROXCINOD
 PubChem
PubChem-
Identification
Product Name:naproxcinod
Synonyms:Naproxcinod;2-(S)-(6-methoxy-2-naphthyl)-propanoic acid 4-nitroxybutyl ester;HCT-3012;2-(S)-(6-methoxy-2-naphthyl)-propanoic acid nitrooxybutyl ester;4-nitrooxybutyl (2S)-2-(6-methoxynaphthalen-2-yl)propanoate;Nitronaproxen;2-(S)-(6-methoxy-2-naphthyl)propanoic acid 4-(nitrooxy)butyl ester;AZD-3582;(S)-2-(6-methoxy-2-naphthyl)propionic acid 4-nitrooxybutyl ester;naproxen-n-butyl nitrate
CAS:163133-43-5
Molecular Formula:C18H21NO6
Molecular Weight:347.36200
Properties
Melting point:N/A
Boiling point:489.475ºC at 760 mmHg
Flash point:193.208ºC
Density:1.213
Stability:
Water solubility:
Index of Refraction:
PSA:90.58000
LogP:4.00680
Vapour pressure:
Safety Data
RIDADR:
Packing group:
HS code:
Request PRICING,IR,NMR,GC,HPLC Chart,COA,MSDS or Route of Synthesis for this product?
Not the right product you are looking for? Contact us for your demand.
Inquire Now- Naproxcinod (Concept Id: C1999704)
- Efficacy safety and effects on blood pressure of naproxcinod 750 mg twice daily compared with placebo and naproxen 500 mg twice daily in patients with osteoarthritis of the hip: a randomized double-blind parallel-group multicenter study.
- Chemistry:Naproxcinod - HandWiki
- Naproxcinod (nitronaproxen) is a nonsteroidal anti-inflammatory drug (NSAID) developed by the French pharmaceutical company NicOx. It is a derivative of naproxen with a nitroxybutyl ester to allow it to also act as a nitric oxide (NO) donor. This second mechanism of action makes naproxcinod the first in a new class of drugs the cyclooxygenase inhibiting nitric oxide …
- Nicox: Fera To Investigate Naproxcinod As Potential Covid ...
- Dec 11 2020· Naproxcinod is a non-steroidal anti-inflammatory product candidate originally discovered and developed by Nicox. Nicox and Fera entered into an agreement in December 2015 which granted Fera ...
- NicOx announces second naproxcinod pivotal phase 3 …
- Sep 15 2008· Naproxcinod 750 mg bid comfortably met the secondary endpoint of being statistically non-inferior to naproxen 500 mg bid on these two parameters. Although not a pre-specified secondary endpoint of ...
- FDA provides Complete Response Letter to NicOxs New …
- Jul 22 2010· FDA provides Complete Response Letter to NicOxs New Drug Application for naproxcinod SOPHIA ANTIPOLIS FRANCE Jul 22 2010 (MARKETWIRE via COMTEX) -- www.nicox.com NicOx S.A. (NYSE Euronext Paris:
- FDA Panel Nixes Naproxcinod for Osteoarthritis
- May 12 2010· Naproxcinod is designed to release naproxen and a nitric oxide donating moiety. NicOx believes that these nitric oxide donating properties permit naproxcinod to mitigate the gastrointestinal ...
- Why NicOx Ran Into an FDA Stone Wall With Its Safer ...
- May 13 2010· NicOxs drug naproxcinod is designed to be a safer treatment for arthritis pain. Safer than say Merck s (MRK) Vioxx (rofecoxib) or Pfizer s (PFE) Bextra (valdecoxib) both of which were pulled ...
- Nicox’s Partner Fera Pharmaceuticals to Investigate ...
- Dec 11 2020· Sophia Antipolis France Nicox SA (Euronext Paris: FR0013018124 COX) an international ophthalmology company and Fera Pharmaceuticals a privately-held U.S. specialty pharmaceutical company announced today that Fera will evaluate naproxcinod as a potential adjuvant treatment for patients with COVID-19 infection. Subject to successful completion of the ongoing manufacturing of naproxcinod ...
- Naproxcinod shows significant advantages over naproxen in ...
- Aug 22 2015· Naproxcinod at 30 mg/kg is effective in the mdx mouse model of DMD. Two doses of naproxcinod (10 and 30 mg/kg) were tested in the mdx mouse model to better assess drug efficacy and range of activity.Mdx mice were treated for 7 months with vehicle and a low or high dose of naproxcinod incorporated into the diet starting at 5 weeks of age. The dose of 30 mg/kg of naproxcinod …
- Effects of naproxcinod on blood pressure in patients with ...
- Naproxcinod is a nitric oxide-donating cyclooxygenase inhibitor in development for osteoarthritis (OA). Thus we characterized the effects of naproxcinod on BP in an integrated safety analysis of 3 pivotal trials of patients with OA of the hip or knee involving 2734 patients.
- A Pharmacokinetics and Safety Study of Naproxcinod in ...
- May 08 2008· A Pharmacokinetics and Safety Study of Naproxcinod in Subjects With Impaired Renal Function (HCT3012-X-106) The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Listing a study does not mean it has been evaluated by the U.S. Federal Government.
- Efficacy and Safety Study of Naproxcinod in Subjects With ...
- Jul 19 2007· To assess the efficacy of naproxcinod compared to naproxen 500 mg bid in relieving OA signs and symptoms in subjects with OA of the knee [ Time Frame: 52 weeks ] To evaluate the effect on blood pressure (BP) of both doses of naproxcinod placebo and naproxen 500 mg bid as measured by the office BP monitoring in a population of OA subjects ...
- Naproxcinod - wikidoc
- Naproxcinod (nitronaproxen) is a non-steroidal anti-inflammatory drug (NSAID) developed by the French pharmaceutical company NicOx. It is a derivative of naproxen with a nitroxybutyl ester to allow it to also act as a nitric oxide (NO) donor.
- Naproxcinod - WikiMili, The Best Wikipedia Reader
- May 22 2021· Naproxcinod (nitronaproxen) is a nonsteroidal anti-inflammatory drug (NSAID) developed by the French pharmaceutical company NicOx.It is a derivative of naproxen with a nitroxybutyl ester to allow it to also act as a nitric oxide (NO) donor. This second mechanism of action makes naproxcinod the first in a new class of drugs the cyclooxygenase inhibiting …
- Fera Requests FDA Orphan Drug Designation for …
- Apr 28 2020· Naproxcinod originally developed by Nicox is a non-steroidal anti-inflammatory treatment candidate specifically designed to release nitric oxide (NO) a gas that induces the relaxation and widening of blood vessels.Estimated Reading Time: 3 mins
- Long-term treatment with naproxcinod significantly ...
- We found that naproxcinod treatment at 21 mg/kg resulted in significant improvement in hindlimb grip strength and a 30% decrease in inflammation in the fore- and hindlimbs of mdx mice. Furthermore we found significant improvement in heart function as evidenced by improved fraction shortening ejection fraction and systolic BP.Cited by: 31Publish Year: 2014Author: Kitipong Uaesoontrachoon James L Quinn Kathleen S Tatem Jack H Van Der Meulen Qing Yu Aditi Pha...
- Naproxcinod FDA Approval Status - Drugs.com
- 9 rows· May 13 2010· Naproxcinod FDA Approval Status. FDA Approved: No Generic name: …DATEARTICLEMar 19 2015Nicox Receives Orphan Drug Designation ...Feb 14 2014Nicox to re-focus Naproxcinod on Duchenne ...Apr 4 2012NicOx Provides Update on U.S. Regulatory ...Jul 22 2010FDA provides Complete Response Letter to ...See all 9 rows on www.drugs.com
- Naproxcinod supplier | CasNO.163133-43-5
- Naproxcinod is a non-steroidal anti-inflammatory drug. It is a derivative of naproxen with a nitroxybutyl ester to allow it to also act as a nitric oxide donor. This second mechanism of action makes naproxcinod the first in a new class of drugs the cyclooxygenase inhibiting nitric oxide donators that are hoped to produce similar analgesic ...
- Naproxcinod (HCT 3012) - COX-Inhibiting Nitric Oxide ...
- Naproxcinod (HCT3012) is an entirely new chemical entity which provides balanced inhibition of COX enzymes while also donating nitric oxide (NO) at sites of inflammation. This mode of action may reduce inflammation as NO is known to exert a relaxing effect on endothelial cells.
